
Phase Diagrams of (NH4)2SO4–Al2(SO4)3–H2O Ternary System: Effect of Sulfuric Acid and Its Application in Recovery of Aluminum from Coal Fly Ash | Journal of Chemical & Engineering Data

Solved) - Al(OH)3(s) + H2SO4(aq) Al2(SO4)3(aq) + H2O(l) Express your answer... (1 Answer) | Transtutors

Mass and Molar quantities of Ca(OH2) and Al2(SO4)3 · 18 H2O used in the... | Download Scientific Diagram

SOLVED: The product of this reaction: Al2O3 + H2O Al2(SO4)3 + H2O Al3(SO4)2 + H2O SO4Al2 + H2O Al2SO4 + H2O

OneClass: Mixing 39.0g of Al(OH)3 with an excess of H2SO4 we produce Al2(SO4)3. Using the following e...

Alumminum hydroxide reacts with sulfuric acid as follows: 2Al(OH)3+H2SO4-->Al2(SO4)+6H2O. Which reagent is the limiting reactant when 0.500 mol Al(OH)3 and 0.500 mol H2SO4 are allowed to react? How ma | Homework.Study.com

Phase Diagrams of (NH4)2SO4–Al2(SO4)3–H2O Ternary System: Effect of Sulfuric Acid and Its Application in Recovery of Aluminum from Coal Fly Ash | Journal of Chemical & Engineering Data
Al + KMnO4 + H2SO4 = KHSO4 + Al2(SO4)3 + MnSO4 + H2O KNO3 + FeSO4 + H2SO4 = KHSO4 + Fe2(SO4)3 + NO H2O H2S + K2Cr2O7 + H2SO4 =KHSO4 + Cr2(SO4)3 + S + H2O Balance the equations using ion electron method.


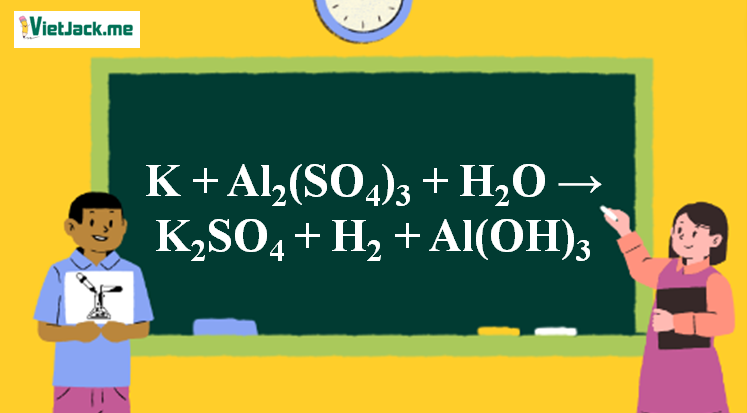



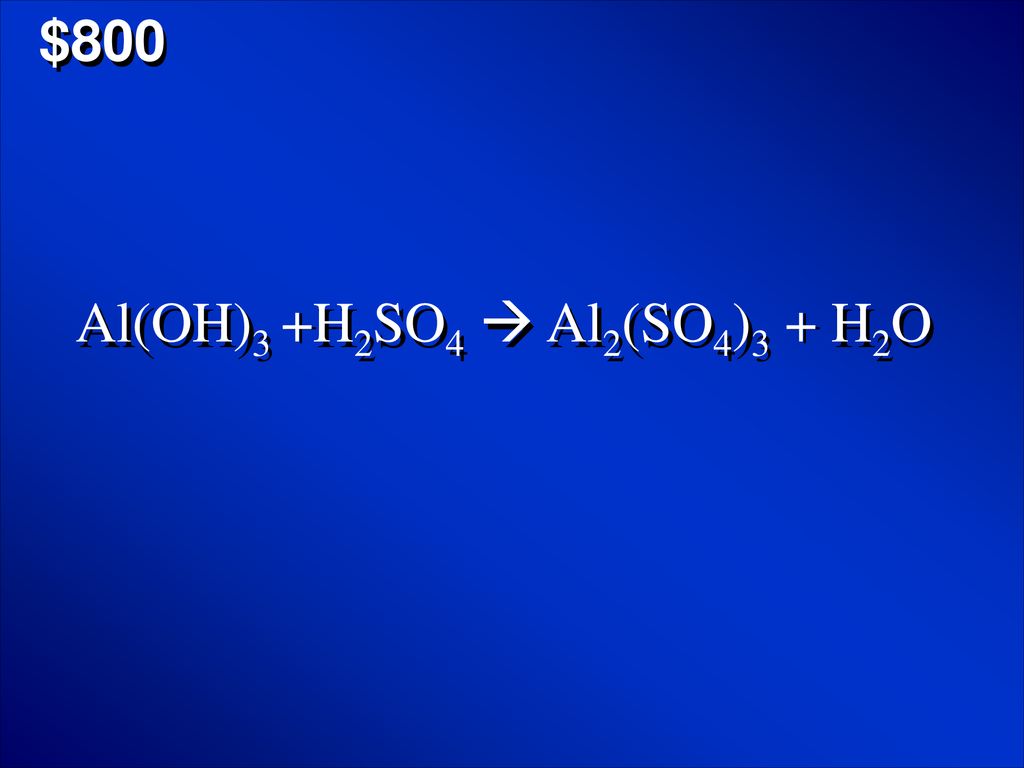




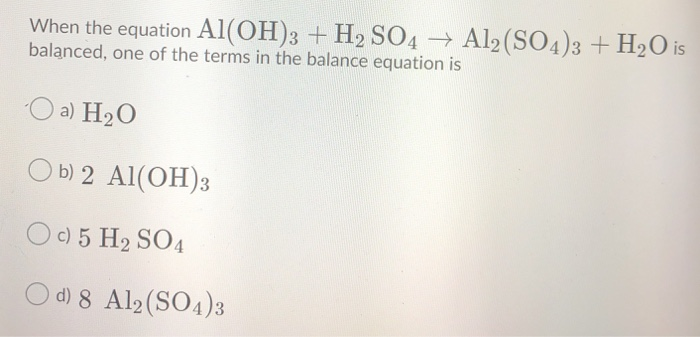

![Aluminium Sulfate Octahydrate [Al2(SO4)3.8H2O] Molecular Weight Calculation - Laboratory Notes Aluminium Sulfate Octahydrate [Al2(SO4)3.8H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/aluminium-sulfate-octahydrate-molecular-weight-calculation-300x178.jpg)



![Punjabi] Balance the chemical equation : Al(OH)3 + H2SO4 → Al2(SO4)3 Punjabi] Balance the chemical equation : Al(OH)3 + H2SO4 → Al2(SO4)3](https://static.doubtnut.com/ss/web-overlay-thumb/10301342.webp)



