Saturation curve showing equilibrium between mole fractions of HAc/H2O... | Download Scientific Diagram

SOLVED: The net ionic equation for the reaction of the weak acid HAc (aq) with the strong base KOH (aq) is: A) H+ (aq) + OH- (aq) â†' H2O B) HAc (aq) +

CV curves of 0.20 mM 4-NP on GCE, MoSe2/GCE, and CeMoSe2/GCE in 0.20 M... | Download Scientific Diagram
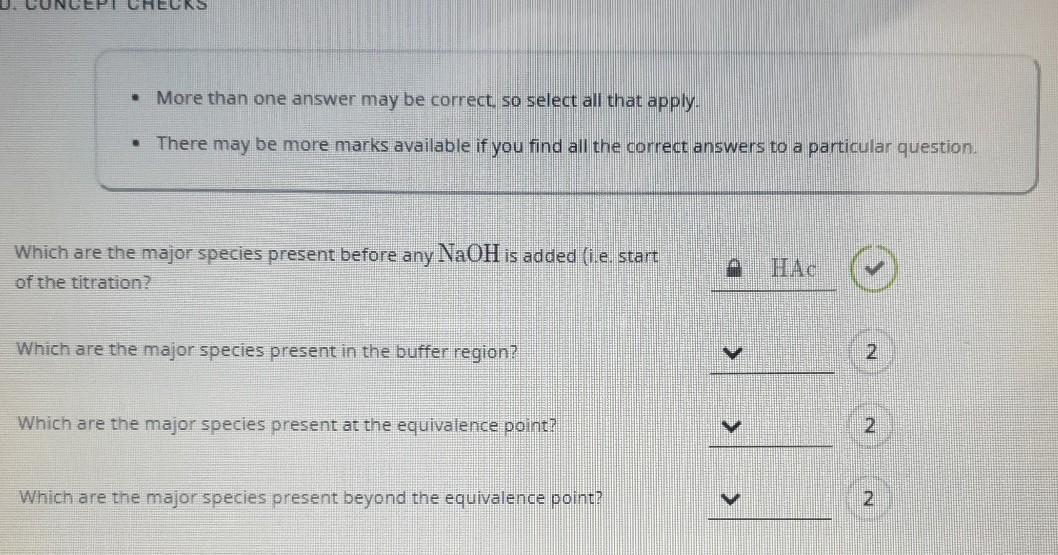
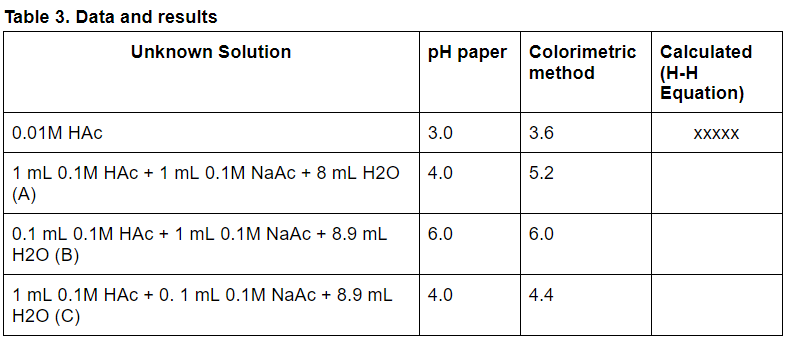
Topic 18- Acids and bases 18.1 Calculations involving acids and bases 18.2 Buffer solutions 18.3 Salt hydrolysis 18.4 Acid-base titrations 18.5 Indicators. - ppt video online download

SEM images of Se nanowires prepared in H2O/HAc mixed solvent at room... | Download Scientific Diagram

SEM images of Se nanowires prepared in H2O/HAc mixed solvent at room... | Download Scientific Diagram

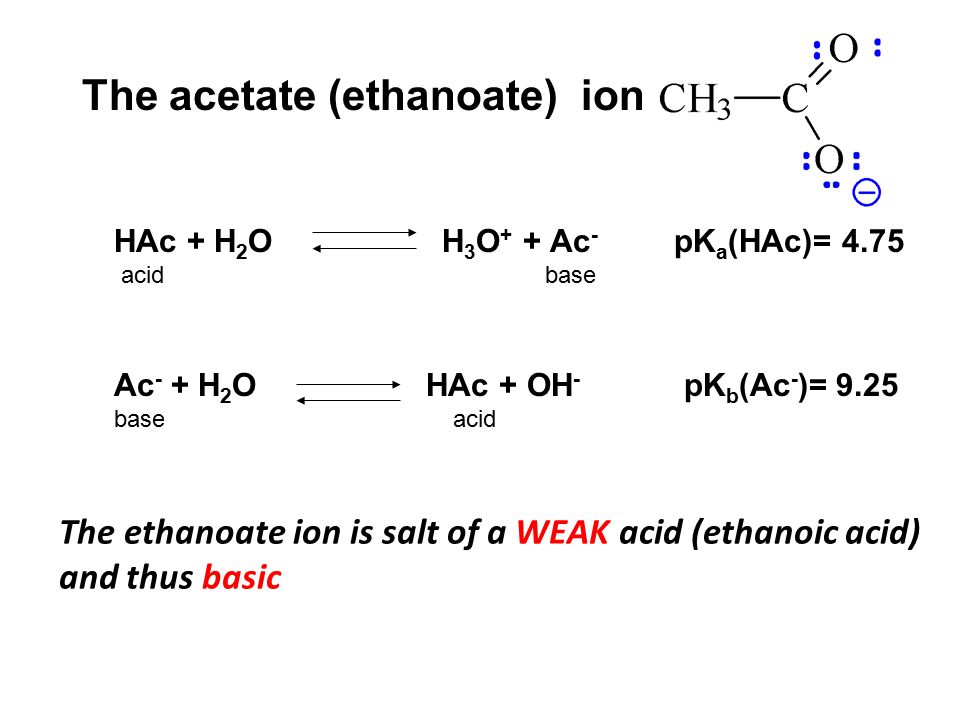
SOLVED: The weak-acid dissociation reaction in water of acetic acid (HAc) to obtain the acetate anion (Ac-) has an equilibrium constant (Keq) of 1.8 x 10^-5 at 298 K. Keq: HAc +
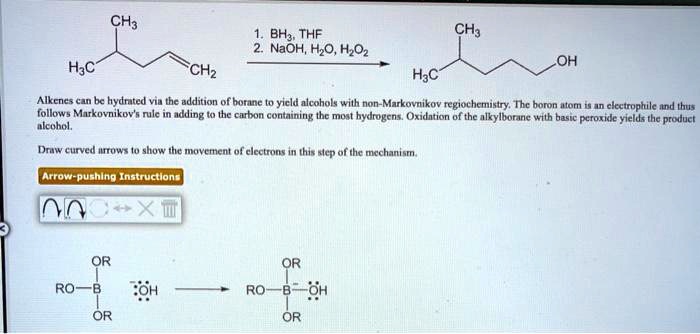
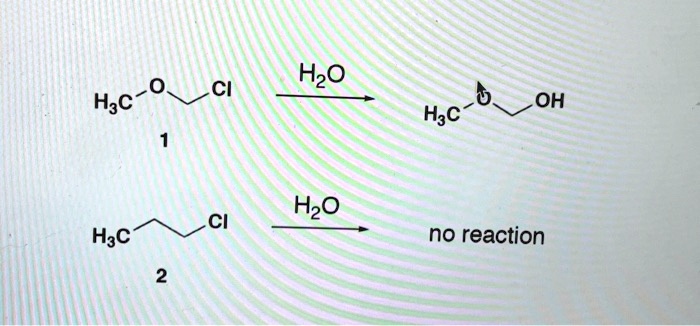
SOLVED: CH3 BHz THF NaOH; H2O, H2O2 HaC CH2 Hac OH Alkenes can be hydrated via the addition of hydrogen to yield alcohols with non-Markovnikov regiochemistry. The boron atom is electrophilic and
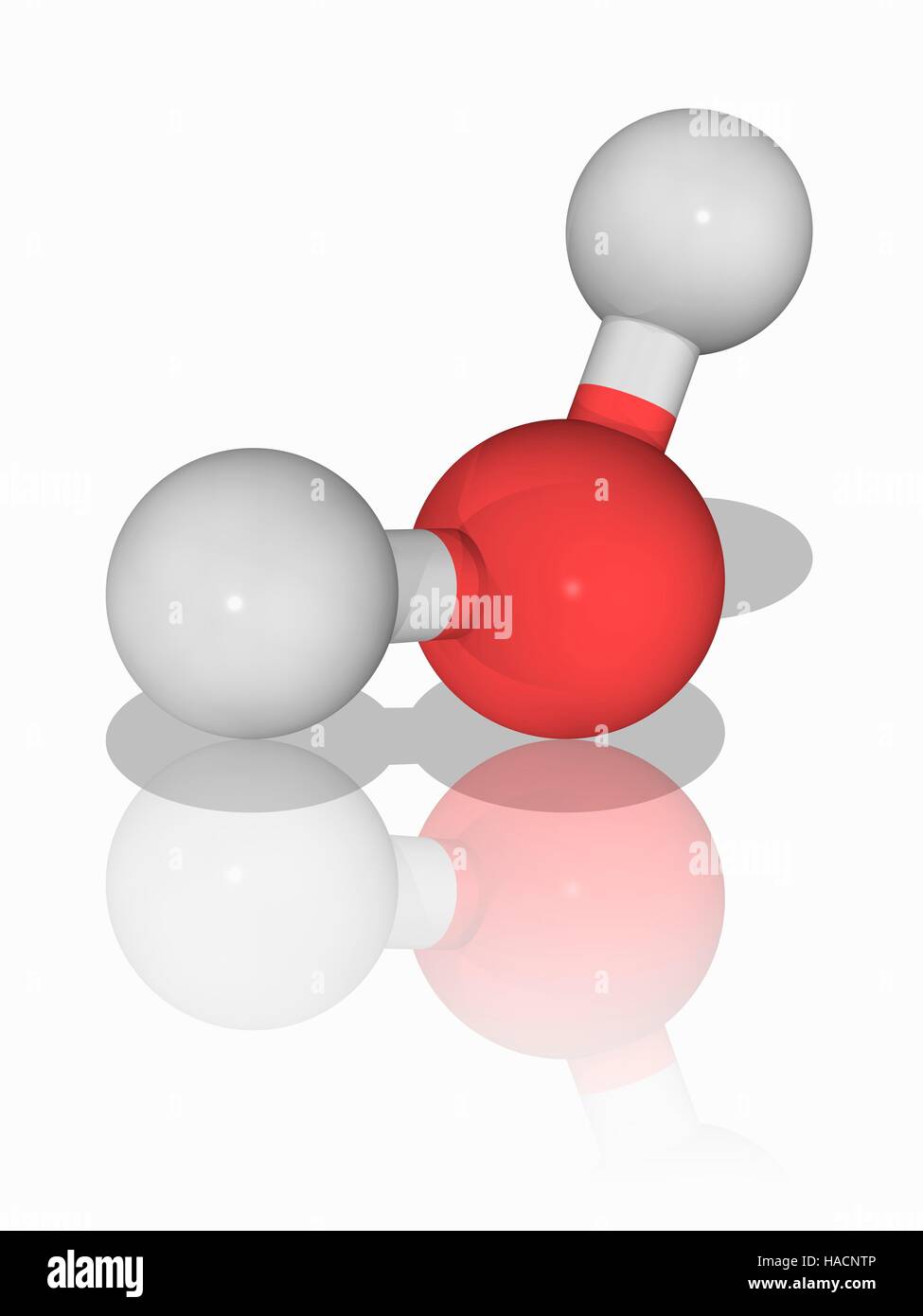
L'eau (H2.O), le modèle moléculaire. Ce liquide est vital pour la vie. L'eau couvre 70 % de la surface de la Terre. Les atomes sont représentés comme des sphères et sont de

SOLVED: Consider the following information: HAc + H2O <=> H3O+ + Ac- Ka = 71.8 x 10^-5 HCN + H2O <=> H3O+ + CN- Ka = 4 x 10^10 What is true?